1 Ocean Acidification
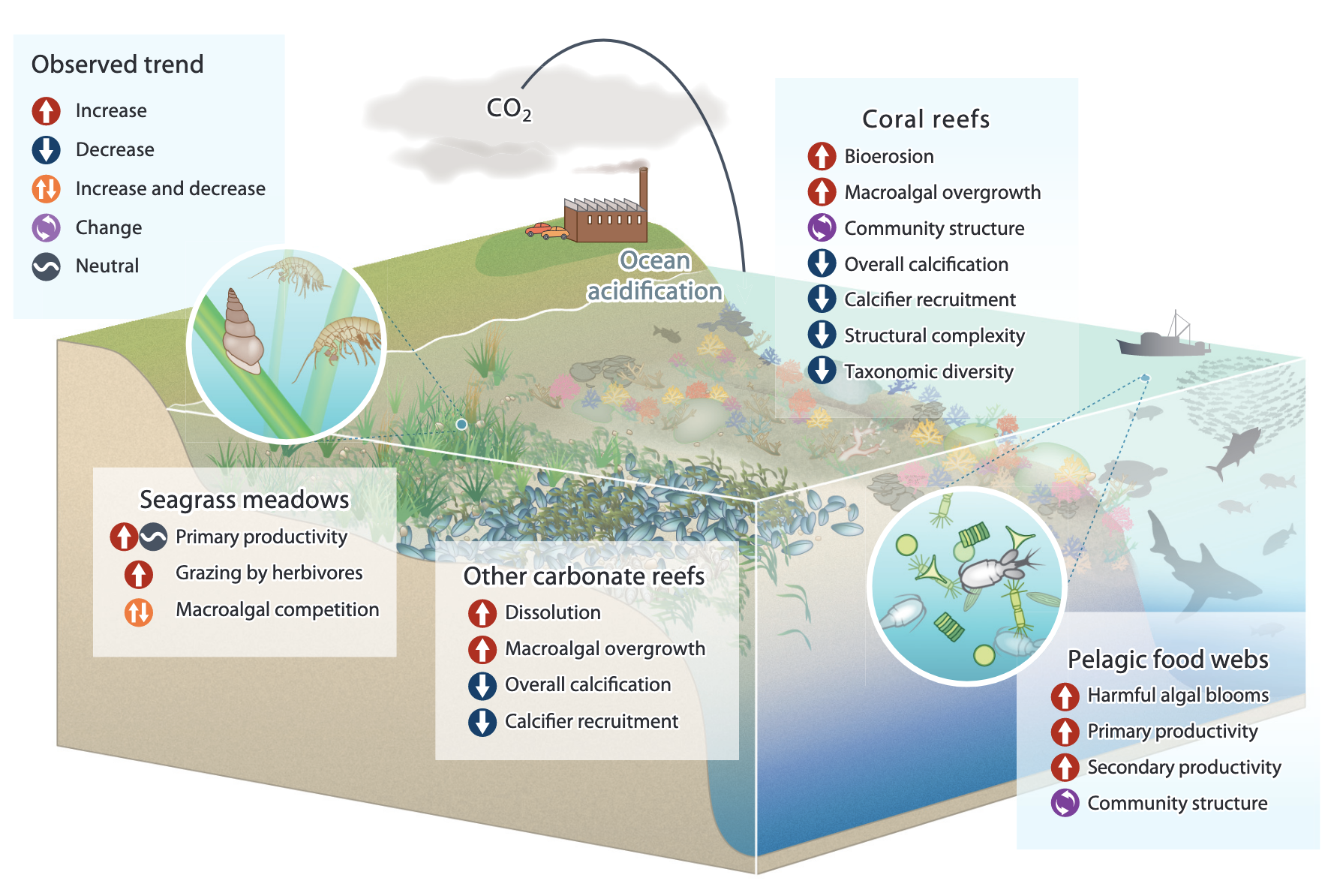
Ocean acidification is one of the most significant threats to coral reefs worldwide. It is a direct consequence of increased atmospheric carbon dioxide (CO₂) levels, which are absorbed by the oceans. This process alters seawater chemistry, with profound impacts not only on coral reefs but on all calcifying marine organisms, their communities and ecosystems
Key papers for this topic include (Doney et al. 2020), (Mollica et al. 2018), (Orr et al. 2005), (Lischka et al. 2025) and (Royal Society 2005).
1.1 Increase in atmospheric carbon dioxide concentration
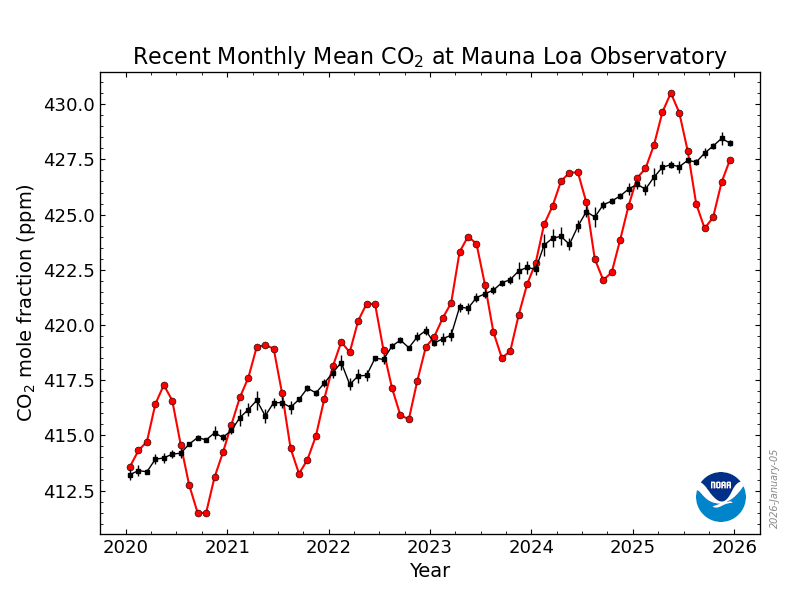
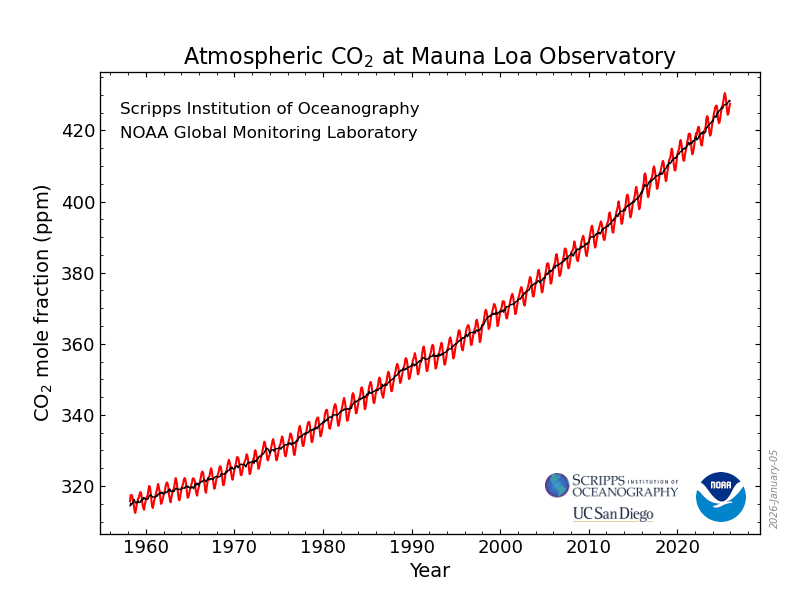
First, we note that atmospheric CO2 levels have risen by about 30% in the last 70 years, from around 300 pm to (now, in 2026) around 428 ppm, and are continuing to rise at a rate of about 2ppm per year. This rate of increase is, if anything, becoming greater. The increase is driven almost entirely by the release into the atmosphere of CO2 in the combustion of fossil fuels. Around 10 Gt carbon is now being released annually. The atmospheric concentration of CO2 is now at a level not experienced on Earth for several million years, and the rate of change in the last two hudred years is up to 100 times greater than the most rapid rates of change that occurred during transitions between glacial and interglacial periods.
The best place to see this is the site of the Mauna Loa Observatory in Hawaii, where measurements of atmospheric CO2 have been going on continually since the late 1950s. From Figure 1.1 we see that over this period there has been an annual cycle superimposed on a relentlessly rising trend.
1.2 Seawater chemistry
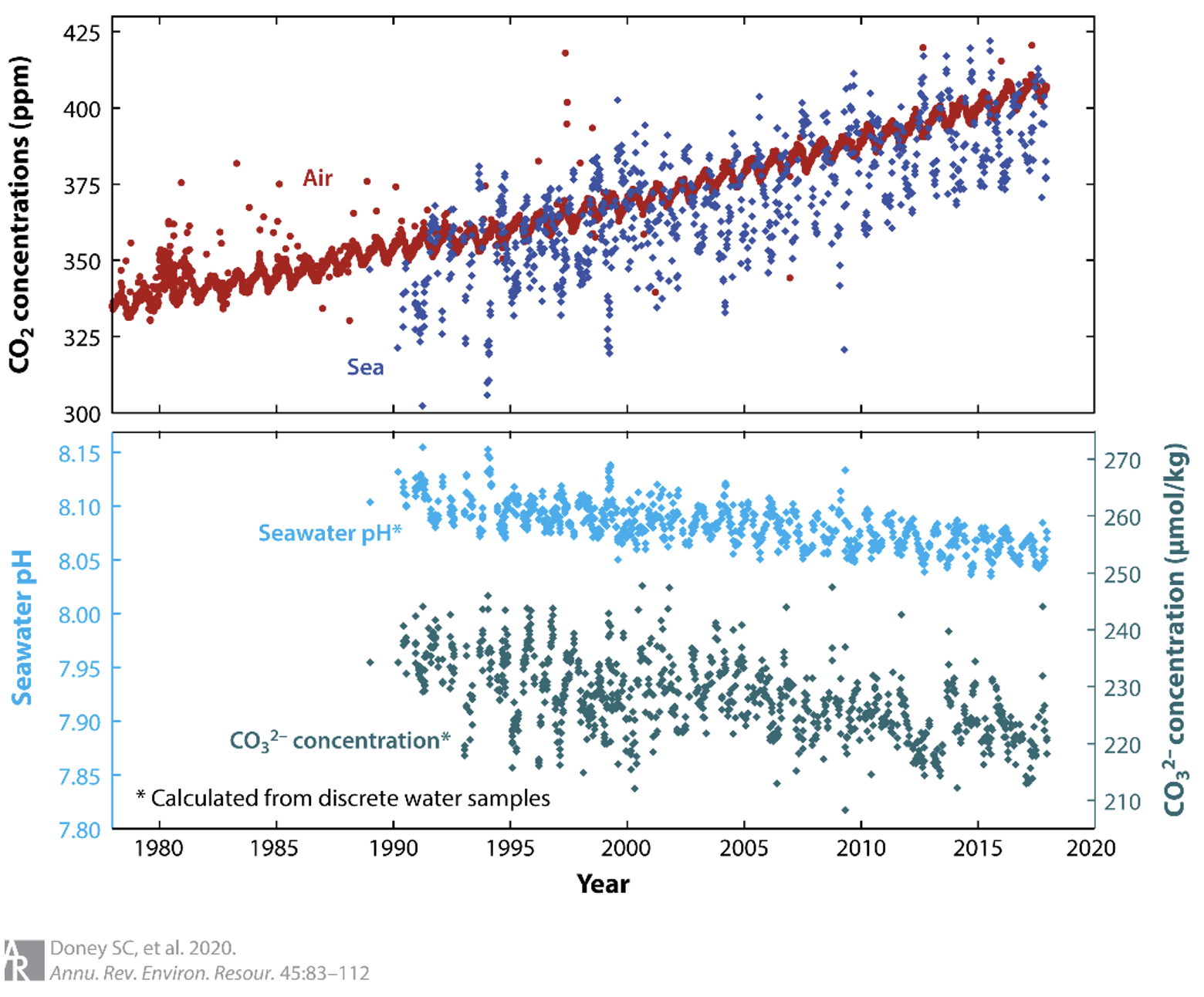
The oceans absorb about 25-30% of the CO₂ emitted into the atmosphere. When CO₂ dissolves in seawater, it reacts with water to form carbonic acid, which dissociates into bicarbonate ions and hydrogen ions. The increase in atmospheric CO2 means that more CO2 is being absorbed by the oceans which means in turn that the concentration of hydrogen ions in the water is increasing. This means that the ocean is becoming more acidic, with reducd pH.
We see from Figure 1.2, taken from (Doney et al. 2020) that ocean pH levels have dropped by about 0.1 over the last 30-40 years.
Let us explore how this happens.
At seawater pH levels (∼8), \(\ce{CO2}\) added to seawater reacts with water to form bicarbonate \(\ce{HCO3-}\) and hydrogen ions \(\ce{H+}\):
\[\ce{CO2 + H2O -> H2CO3- + H+}\] The release of \(\ce{H+}\) acts to increase acidity and lower seawater pH, defined as
\[\ce{pH}=-\log_{10}\ce{[H+]}\]
pH: a measure of the acidity of water where lower pH reflects more acidic conditions; reported on a log-scale such that a 1-unit drop in pH is equivalent to a factor of 10 increase in acidity
and lower the concentration of carbonate ions , via
\[\ce{CO3^{2-} + H+ -> HCO3-}\] Acidification impacts will depend on organism responses to multiple, simultaneous chemical changes—increasing \(\ce{CO2}\), \(\ce{HCO3-}\) , and \(\ce{H+}\) and decreasing \(\ce{CO3^{2-}}\). Many types of marine organisms that form shells and skeletons from calcium carbonate (\(\ce{CaCO3}\)) minerals are sensitive to acidification. The solubility of carbonate minerals,
\[\ce{CaCO3 (s) <-> CO3^{2-} + Ca2+}\]
can be expressed as a carbon saturation state
\[ \Omega = \frac{\ce{[CO3^{2-}][Ca2+]}}{K_{sp}} \]
where \(K_{sp}\) is the apparent solubility product at a given temperature, salinity and pressure for each mineral form of \(\ce{CaCO3}\). A value of \(\Omega\) < 1 indicates undersaturation with respect to thermodynamic equilibrium, and under those seawater conditions, unprotected carbonate materials will dissolve. The multiple forms of carbonate minerals vary in Ksp and so have different solubilities, with calcite being less soluble than aragonite and amorphous calcium carbonate. As \(\ce{CO2}\) increases, the \(\ce{CO3^{2-}}\) concentration declines because of consumption with \(\ce{H+}\) (Equation 3) causing a decline in \(\Omega\).
aragonite and calcite are two mineral forms of calcium carbonate \(\ce{CaCO3}\). Both are used by marine organisms in shell and skeleton formation by a biominerlization. Aragonite is the more soluble of the two, with rapid dissolution kinetics
Ocean surface waters exchange \(\ce{CO2}\) with the overlying atmosphere via physical gas transfer, and the surface seawater partial pressure, \(\ce{pCO2}\), tends to track the growth of atmospheric \(\ce{CO2}\) for much of the global ocean, as illustrated by long-term time series records at numerous open-ocean locations and analysis of global surface ocean \(\ce{CO2}\) observational networks. As a result, surface pH and \(\ce{CO3^{2-}}\) are declining, and surface ocean pH is estimated to have dropped on average globally by approximately 0.1 units from the preindustrial era to present, which means there has been an ∼30% increase in hydrogen ion concentration. What? Yes. The oceans are now about 30% more acidic than they were just a few decades ago.
Hence, as \(\Omega\) declines, aragonite in particular becomes more soluble and aragonite calcareous shells of marine life forms become vulnerable to dissolving.
Studies have already found evidence of extensive shell dissolution of the aragonite shells of the pteropod (small snails) Limacina helicina antarctica in the Southern Ocean, where undersaturation (ie \(\ce{Omega_{\text{ar}}<1})\) of aragonite is now a reality (Lischka et al. 2025).
1.3 Organismal Responses
Changes to
- growth of otoliths—calcium carbonate structures in fish ears that aid in balance—mitochondrial function
- metabolic rate
- larval yolk consumption
- activity
- neurosensory processes
- behaviour, including settlement into specific habitat at the end of the early life stages.
Variable responses - must take into account interactions with other factors such s elevted temperature and depleted oxygen.
1.4 Community and Ecosystem Effects